China Cosmetics Safety and Efficacy Evaluation Requirements

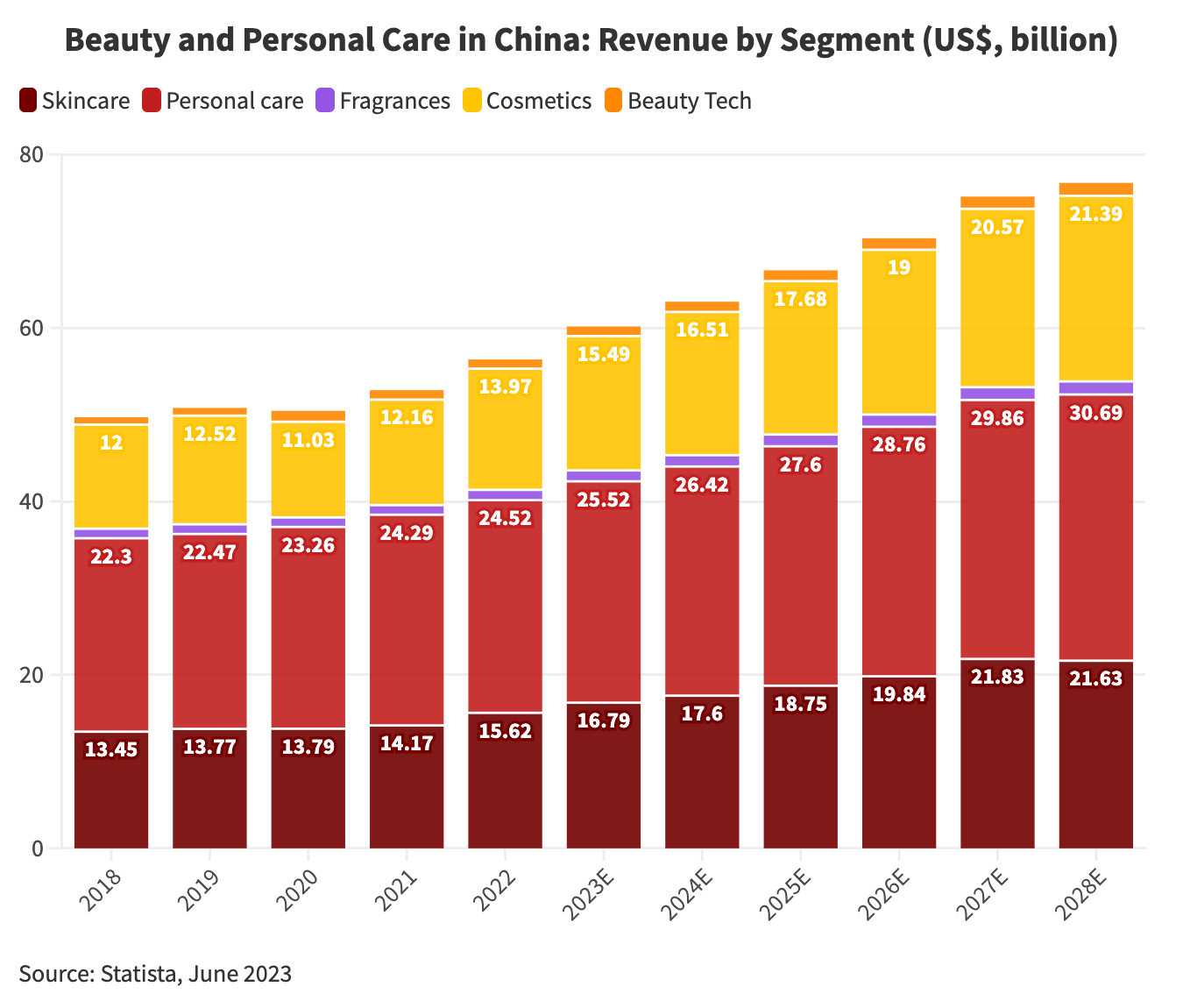
As per the Cosmetics Supervision and Administration Regulations (CSAR), cosmetics efficacy claims must be supported by a solid scientific foundation. Cosmetic registrants and filers are required to submit a summary of research data or product efficacy evaluations to the NMPA-designated platform for public review and monitoring.
The registrants and filers are responsible for ensuring that the submitted summary is scientifically accurate, reliable, and traceable.
Exemptions from Efficacy Evaluation
Certain cosmetic products do not require efficacy evaluation. These include products such as:
- Products for cleaning, makeup removal, beauty and modification, fragrance, refreshing, hair dyeing, hair perming, hair color care, depilation, deodorization, and auxiliary shaving.
- Whitening products, exfoliators, and deep pore cleansers (when labeled as having physical effects only).
Efficacy Evaluation Requirements

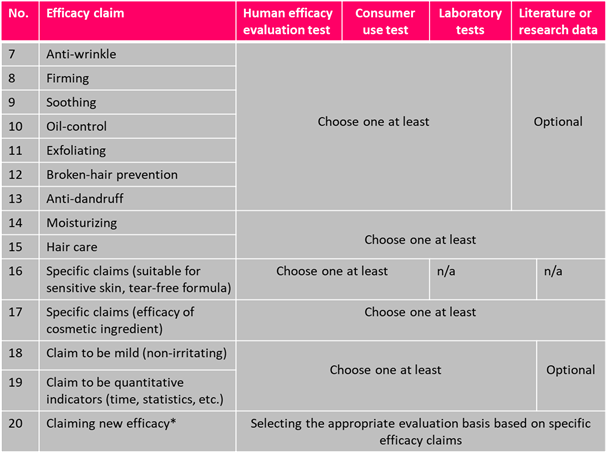
Transitional Solutions
|
Cosmetic efficacy claims must be evaluated according to the Specification for the Evaluation of Cosmetic Efficacy Claims, and the efficacy data summary must be uploaded to the NMPA platform. |
New Cosmetic Registration and Filing |
Cosmetics Approved or Filed Before May 1, 2021 | Cosmetics Approved or Filed Between May 1, 2021 and Dec 31, 2021 |
|---|---|---|---|
| Since 2022 | Before May 1, 2023 | Before May 1, 2022 |
Our Expert Services
Further Information
For additional details on testing and evaluation services, we offer an in-vitro human hair testing service to assist with your cosmetic product development.